CT as gatekeeper of invasive coronary angiography in patients with suspected CAD
Introduction
Coronary artery disease (CAD) continues to be a major cause of mortality and disability in the 21 century (1). The disease often manifests itself with chest pain but could be fatal even in its first manifestation and occur without warning signals in asymptomatic subjects. The clinician’s desire to avoid missing clinically-relevant CAD explains the recent surge in usage of advanced medical imaging. However, this “defensive” approach did not translate into a reduction in cardiac events (2). A cross-sectional, population-based sample of Medicare patients from 1993 to 2001 found that overall hospitalizations for acute myocardial infarction remained flat at 8.7 per 1,000 patients despite significant increases in imaging and revascularization rates (3). The clinician nowadays is equipped with an increasing number of tests for the assessment of individuals who have symptoms suggestive of CAD or for risk-stratifying the asymptomatic individual. However, these tests come with a cost and financial constraints in the present economic environment will no longer allow its indiscriminate use. The gatekeeper function of a diagnostic testing strategy implies that a test is selected judiciously with the aim of preventing access to invasive yet expensive coronary angiography. An effective gatekeeper function is defined when, after the test is performed, patient management is promptly targeted by the noninvasive test findings without the need for additional diagnostic procedures (4,5).
In accordance with current practice guidelines, the diagnostic work-up of suspected CAD is still mainly advocated by the results of noninvasive stress tests (6,7). With the emergence of computed tomography coronary angiography (CTCA) noninvasive access to coronary anatomy has become available and offers a viable alternative to the traditional workup using functional tests. Noninvasive imaging now unravels coronary anatomy in many cases, including asymptomatic subjects, who would normally not undergo invasive coronary angiography (ICA) and changes the gatekeeper function to the cathlab that until very recently was exclusively reserved to noninvasive functional imaging.
CTCA as a robust diagnostic and prognostic test
The diagnostic performance of CTCA in comparison with ICA has been demonstrated in numerous single-center studies and three prospective multicenter trials (8-11). CTCA has a demonstrated 94% to 99% sensitivity and 64% to 83% specificity across a range of disease prevalence and inclusive of patients with both acute and stable chest pain. The 97% to 99% negative predictive value means that CTCA can effectively rule out anatomically significant CAD, which is defined as the presence of at least one 50% or greater coronary artery stenosis.
The prognostic value of CTCA has also been extensively studied (12,13). What becomes clear from these data is that subjects with signs of CAD, also in its early stages, are at higher risk than those who have no coronary plaque. Individuals without evidence of CAD by CTCA have a benign outcome, with annualized cardiac event rates of 0.01% to 0.24%, and this for a long-term period of at least 5 years (14). On the other hand, whenever CAD is present, data from several large clinical registries consistently demonstrate a risk continuum of adverse events with the extent of atherosclerotic disease, without a threshold effect for lumen obstruction or hemodynamically significant CAD (12).
Nonobstructive coronary atherosclerosis: redefining the criteria for CAD and implications for management
Recent clinical practice guidelines support the use of CTCA as a primary or secondary diagnostic option in symptomatic patients with an intermediate likelihood of CAD (6,7). Accordingly, when using CTCA as an up-front diagnostic test, atherosclerosis is often detected during its early stages in “healthy” carriers of the disease or subjects with mild or atypical symptoms. This new terminology of obstructive and nonobstructive plaque challenges existing clinical practice. Indeed, patients who have evidence of CAD, but without inducible ischemia or significant stenosis, are considered low risk for cardiovascular death, and the appropriate use of preventive measures is currently not endorsed by practice guidelines (15). As a result, secondary prevention is less frequently implemented in these patients. However, the demonstration of nonobstructive CAD by means of CTCA may prove to be a valuable tool for the appropriate allocation of preventive therapies such as statin and aspirin. In a recent study, statin therapy was associated with a significantly lower mortality for individuals with atherosclerotic plaque on CTCA, but not for individuals with normal coronary arteries (16). Symptomatic patients assigned to a CTCA strategy in the Prospective Multicenter Imaging Study for Evaluation of Chest Pain (PROMISE) and Scottish Computed Tomography of the Heart (SCOT-HEART) trials were found to have a trend towards lower rates of myocardial infarction at follow-up than those randomized to functional testing, which is attributed to greater utilization of secondary preventive measures in response to visualizing (mostly nonobstructive) CAD (17,18).
These new insights make it necessary to redefine the criteria to establish the diagnosis of CAD, which date back 50 years when coronary atherosclerosis could only be detected by ICA (19). The old paradigm defined disease based on the simple categorization of presence or absence of obstructive CAD, using the criterium of 50% or greater diameter stenosis by ICA. In the new paradigm, risk from CAD does not abruptly increase with the presence of a stenosis, but reflects the burden of disease on a wide spectrum (20). The risk of myocardial infarction or cardiovascular death is exceedingly low unless disease is detectable by CTCA (21). Patients with limited nonobstructive disease have worse prognosis than patients with normal coronary arteries, but do better than subjects with extensive nonobstructive disease. In patients with extensive nonobstructive disease, risk equals those with single-vessel obstructive CAD. In this newly proposed CAD classification scheme suitable for both conventional and CT angiography assessment, the extent of CAD was classified in stages as follows: stage 0, no coronary atherosclerotic disease by coronary angiography; stage 1, mild coronary atherosclerotic disease: <30% lumen stenosis affecting 1 or 2 vessels; stage 2, moderate coronary atherosclerotic disease: 30–49% lumen stenosis affecting 1 or 2 vessels or mild disease in 3 vessels; stage 3, severe coronary atherosclerotic disease: ≥50% lumen stenosis affecting 1 or 2 vessels or moderate disease in 3 vessels; and stage 4, very severe coronary atherosclerotic disease: ≥50% lumen stenosis affecting 3 vessels, or 2 vessels including proximal LAD, or left main disease. Such CAD stages were directly related to an increasing annual risk of myocardial infarction or cardiovascular death (stage 0: <0.1%; stage 1: 0.1–0.9%; stage 2: 1–1.9%; stage 3: 2–4%, and stage 4: >4%) (20).
Which diagnostic test to use in clinical practice?
Cardiac CT has been accepted as a valuable diagnostic tool in today’s patient care. However, several other noninvasive, in particular, functional diagnostic tests are available on the menu for the ordering clinician and target more or less the same patient population. In the present economic environment with limited budgets, the cost-effectiveness of a diagnostic strategy comes into play and it therefore becomes essential to select the appropriate diagnostic test. Several randomized clinical trials have been conducted to evaluate the health outcomes of patients in need of a diagnostic work-up comparing a CT-based anatomic approach with traditional functional tests (17,18,22,23). It turns out that the choice of the diagnostic test does not result in a difference in clinical outcome, leading to the conclusion that a CT-based anatomical and functional strategy is comparable. Pooling of data from these 14,817 patients in a meta-analysis demonstrated two salient features (24). First, selecting cardiac CT as a default strategy was associated with lower rates of myocardial infarction at follow-up than those randomized to functional testing, which is attributed to greater utilization of secondary prevention measures in response to visualizing (mostly nonobstructive) CAD. Second, choosing CTCA as the initial test increased the rate of revascularizations as well as a trend toward more ICAs. Data from another multicentre randomized study, the CRESCENT trial, demonstrated that the use of cardiac CT in clinical practice was at least as effective as the traditional approach using functional testing, but with less downstream diagnostic testing and thus lower cumulative diagnostic costs when following these patients for more than 1 year (25).
The potential for overuse of ICA and revascularizations when using CTCA instead of functional testing is of concern both from a patient (risk of harm in low-risk individuals) and society (higher costs) perspective and has been reported in several studies (24,26). Unfamiliarity with the technique, both its possibilities and limitations, is one possible explanation. More importantly, the practice to directly refer patients with CAD on CTCA for invasive confirmation, finds it origin in the tendency of CTCA to overestimate stenosis severity, or in other words its low positive predictive value (10). In addition, catheter-based confirmation of a significant stenosis on CTCA is often not enough for adequate patient management (27). Once CAD has been identified on CTCA (or ICA), documentation of the ischemic potential of a stenosis becomes important to guide patient therapy (28). Patients with anatomically proven CAD but no evidence of ischemia do well with drug therapy (such as statins and antiplatelet drugs) only. Patients with anatomical disease and evidence of ischemia are in general better off with revascularisation on top of drug therapy (29). Hence, in clinical practice both anatomic and functional data often will be necessary (Figure 1). This awareness has been the trigger to assess the functional significance of anatomical disease as detected by CTCA, using different modalities, including the exercise treadmill, single-photon emission computed-tomography, positron emission tomography and CT myocardial perfusion imaging (30-34). One particular and relatively new approach, is the calculation of fractional flow reserve (FFR) from standard CTCA acquisitions using computational fluid dynamics (FFRCT) (35-37). This method allows to calculate FFR at any point in the coronary vascular bed, without the need for additional imaging, modification of image acquisition protocols, additional radiation doses, or medication administration. Preliminary data show that the addition of functional information, using FFRCT data, enables to cut down on the use of ICA, the main driver of costs within the area of CAD (38).
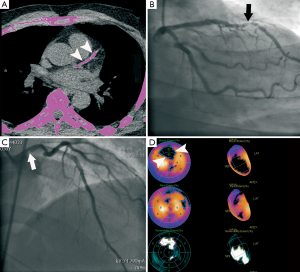
CTCA as gatekeeper to ICA?
Although guidelines recommend the selective referral to ICA based on the results of prior noninvasive stress testing, this approach is not widespread. Currently up to one-third of nonemergent ICAs are being performed in the absence of any sort of noninvasive functional evaluation (39). Even when stress testing is applied more rigorously, up to 40% of ICAs detect normal coronary arteries (40). In a recent study, comparing different noninvasive cardiac imaging tests as gate-keeper versus upfront ICA, ICA was still performed in about 80% of patients in the noninvasive groups (41). The probable explanation is that the use of ICA is driven by ongoing symptoms, in spite of having a normal stress test result. In other words, the characterisation of a patient as low risk is often insufficient to allay concerns of the clinician and/or patient. As a corollary, new strategies are needed to increase the diagnostic yield of cardiac catheterisation in routine clinical practice (40).
Cardiac CT may effectively fill this need as it reliably provides the clinician with essential information, previously unavailable when using noninvasive stress tests. As already alluded to, CTCA easily identifies individuals with nonobstructive CAD, who are at risk of incident myocardial infarction and ischemic cardiac death, and could therefore help to implement effective preventive measures (e.g., statin therapy) in patients with (subclinical) evidence of CAD (16). Unlike stress testing, CTCA is also reliable for the identification and exclusion of high-risk (left main or 3-vessel) CAD (42,43). Real-world data provide ammunition to the view that CTCA serves as an effective gatekeeper to ICA, by demonstrating a reduction in the use of ICA as low as 12% over a 2-year post-test period (5,44).
Conclusions
Over the past two decades CTCA has matured into a valuable diagnostic test in today’s patient care. However, many other noninvasive tests are available for assessing the individual with possible CAD and it is anticipated that the emphasis will be placed more than ever on cost-effectiveness of diagnostic testing strategies. The use of CTCA as gatekeeper to ICA looks promising and is further being tested in prospective multicenter studies. Information obtained from functional tests will remain necessary to guide referral for ICA and coronary revascularisation decisions. In any case, it has become clear that the emergence of CTCA has changed existing clinical practice and will have a central role in the future referral pattern of patients to the cathlab. Notably in this respect is the design of the ongoing ISCHEMIA trial (ClinicalTrials.gov number NCT01471522). This prospective large-scale study randomizes 8000 patients with ischemia to an invasive strategy in addition to optimal medical therapy (OMT) versus a non-invasive approach of OMT, with catheterisation and possible revascularisation being reserved for those patients whose medical therapy fails. Acknowledging the limitations of stress testing, all patients will undergo CTCA before ICA to exclude significant left main CAD and confirm obstructive CAD prior to randomization.
Acknowledgements
None.
Footnote
Conflicts of Interest: The author has no conflicts of interest to declare.
References
- Go AS, Mozaffarian D, Roger VL, et al. Heart disease and stroke statistics--2014 update: a report from the American Heart Association. Circulation 2014;129:e28-e292. [Crossref] [PubMed]
- Redberg RF, Walsh J. Pay now, benefits may follow--the case of cardiac computed tomographic angiography. N Engl J Med 2008;359:2309-11. [Crossref] [PubMed]
- Lucas FL, DeLorenzo MA, Siewers AE, et al. Temporal trends in the utilization of diagnostic testing and treatments for cardiovascular disease in the United States, 1993-2001. Circulation 2006;113:374-9. [Crossref] [PubMed]
- Shaw LJ, Hachamovitch R, Berman DS, et al. The economic consequences of available diagnostic and prognostic strategies for the evaluation of stable angina patients: an observational assessment of the value of precatheterization ischemia. Economics of Noninvasive Diagnosis (END) Multicenter Study Group. J Am Coll Cardiol 1999;33:661-9. [Crossref] [PubMed]
- Shaw LJ, Hausleiter J, Achenbach S, et al. Coronary computed tomographic angiography as a gatekeeper to invasive diagnostic and surgical procedures: results from the multicenter CONFIRM (Coronary CT Angiography Evaluation for Clinical Outcomes: an International Multicenter) registry. J Am Coll Cardiol 2012;60:2103-14. [Crossref] [PubMed]
- Task Force Members. 2013 ESC guidelines on the management of stable coronary artery disease: the Task Force on the management of stable coronary artery disease of the European Society of Cardiology. Eur Heart J 2013;34:2949-3003. [Crossref] [PubMed]
- Fihn SD, Gardin JM, Abrams J, et al. 2012 ACCF/AHA/ACP/AATS/PCNA/SCAI/STS Guideline for the diagnosis and management of patients with stable ischemic heart disease: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, and the American College of Physicians, American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J Am Coll Cardiol 2012;60:e44-e164. [Crossref] [PubMed]
- von Ballmoos MW, Haring B, Juillerat P, et al. Meta-analysis: diagnostic performance of low-radiation-dose coronary computed tomography angiography. Ann Intern Med 2011;154:413-20. [Crossref] [PubMed]
- Miller JM, Rochitte CE, Dewey M, et al. Diagnostic performance of coronary angiography by 64-row CT. N Engl J Med 2008;359:2324-36. [Crossref] [PubMed]
- Meijboom WB, Meijs MF, Schuijf JD, et al. Diagnostic accuracy of 64-slice computed tomography coronary angiography: a prospective, multicenter, multivendor study. J Am Coll Cardiol 2008;52:2135-44. [Crossref] [PubMed]
- Budoff MJ, Dowe D, Jollis JG, et al. Diagnostic performance of 64-multidetector row coronary computed tomographic angiography for evaluation of coronary artery stenosis in individuals without known coronary artery disease: results from the prospective multicenter ACCURACY (Assessment by Coronary Computed Tomographic Angiography of Individuals Undergoing Invasive Coronary Angiography) trial. J Am Coll Cardiol 2008;52:1724-32. [Crossref] [PubMed]
- Min JK, Dunning A, Lin FY, et al. Age- and sex-related differences in all-cause mortality risk based on coronary computed tomography angiography findings results from the International Multicenter CONFIRM (Coronary CT Angiography Evaluation for Clinical Outcomes: An International Multicenter Registry) of 23,854 patients without known coronary artery disease. J Am Coll Cardiol 2011;58:849-60. [Crossref] [PubMed]
- Bittencourt MS, Christman MP, Hulten E, et al. Comparison of the use of downstream tests after exercise treadmill testing by cardiologists versus noncardiologists. Am J Cardiol 2014;114:305-11. [Crossref] [PubMed]
- Pontone G, Andreini D, Bartorelli AL, et al. A long-term prognostic value of CT angiography and exercise ECG in patients with suspected CAD. JACC Cardiovasc Imaging 2013;6:641-50. [Crossref] [PubMed]
- Smith SC Jr, Benjamin EJ, Bonow RO, et al. AHA/ACCF secondary prevention and risk reduction therapy for patients with coronary and other atherosclerotic vascular disease: 2011 update: a guideline from the American Heart Association and American College of Cardiology Foundation endorsed by the World Heart Federation and the Preventive Cardiovascular Nurses Association. J Am Coll Cardiol 2011;58:2432-46. [Crossref] [PubMed]
- Chow BJ, Small G, Yam Y, et al. Prognostic and therapeutic implications of statin and aspirin therapy in individuals with nonobstructive coronary artery disease: results from the CONFIRM (COronary CT Angiography EvaluatioN For Clinical Outcomes: An InteRnational Multicenter registry) registry. Arterioscler Thromb Vasc Biol 2015;35:981-9. [Crossref] [PubMed]
- Douglas PS, Hoffmann U, Patel MR, et al. Outcomes of anatomical versus functional testing for coronary artery disease. N Engl J Med 2015;372:1291-300. [Crossref] [PubMed]
- SCOT-HEART investigators. CT coronary angiography in patients with suspected angina due to coronary heart disease (SCOT-HEART): an open-label, parallel-group, multicentre trial. Lancet 2015;385:2383-91. [Crossref] [PubMed]
- Sones FM Jr. Indications and value of coronary arteriography. Circulation 1972;46:1155-60. [Crossref] [PubMed]
- Arbab-Zadeh A, Fuster V. The Risk Continuum of Atherosclerosis and its Implications for Defining CHD by Coronary Angiography. J Am Coll Cardiol 2016;68:2467-78. [Crossref] [PubMed]
- Habib PJ, Green J, Butterfield RC, et al. Association of cardiac events with coronary artery disease detected by 64-slice or greater coronary CT angiography: a systematic review and meta-analysis. Int J Cardiol 2013;169:112-20. [Crossref] [PubMed]
- McKavanagh P, Lusk L, Ball PA, et al. A comparison of cardiac computerized tomography and exercise stress electrocardiogram test for the investigation of stable chest pain: the clinical results of the CAPP randomized prospective trial. Eur Heart J Cardiovasc Imaging 2015;16:441-8. [Crossref] [PubMed]
- Min JK, Koduru S, Dunning AM, et al. Coronary CT angiography versus myocardial perfusion imaging for near-term quality of life, cost and radiation exposure: a prospective multicenter randomized pilot trial. J Cardiovasc Comput Tomogr 2012;6:274-83. [Crossref] [PubMed]
- Bittencourt MS, Hulten EA, Murthy VL, et al. Clinical Outcomes After Evaluation of Stable Chest Pain by Coronary Computed Tomographic Angiography Versus Usual Care: A Meta-Analysis. Circ Cardiovasc Imaging 2016;9:e004419. [Crossref] [PubMed]
- Lubbers M, Dedic A, Coenen A, et al. Calcium imaging and selective computed tomography angiography in comparison to functional testing for suspected coronary artery disease: the multicentre, randomized CRESCENT trial. Eur Heart J 2016;37:1232-43. [Crossref] [PubMed]
- Shreibati JB, Baker LC, Hlatky MA. Association of coronary CT angiography or stress testing with subsequent utilization and spending among Medicare beneficiaries. JAMA 2011;306:2128-36. [Crossref] [PubMed]
- Meijboom WB, Van Mieghem CA, van Pelt N, et al. Comprehensive assessment of coronary artery stenoses: computed tomography coronary angiography versus conventional coronary angiography and correlation with fractional flow reserve in patients with stable angina. J Am Coll Cardiol 2008;52:636-43. [Crossref] [PubMed]
- Task Force on Myocardial Revascularization of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery. Guidelines on myocardial revascularization. Eur Heart J 2010;31:2501-55. [Crossref] [PubMed]
- Hachamovitch R, Hayes SW, Friedman JD, et al. Comparison of the short-term survival benefit associated with revascularization compared with medical therapy in patients with no prior coronary artery disease undergoing stress myocardial perfusion single photon emission computed tomography. Circulation 2003;107:2900-7. [Crossref] [PubMed]
- Nieman K, Galema T, Weustink A, et al. Computed tomography versus exercise electrocardiography in patients with stable chest complaints: real-world experiences from a fast-track chest pain clinic. Heart 2009;95:1669-75. [Crossref] [PubMed]
- Acampa W, Gaemperli O, Gimelli A, et al. Role of risk stratification by SPECT, PET, and hybrid imaging in guiding management of stable patients with ischaemic heart disease: expert panel of the EANM cardiovascular committee and EACVI. Eur Heart J Cardiovasc Imaging 2015;16:1289-98. [Crossref] [PubMed]
- Flotats A, Knuuti J, Gutberlet M, et al. Hybrid cardiac imaging: SPECT/CT and PET/CT. A joint position statement by the European Association of Nuclear Medicine (EANM), the European Society of Cardiac Radiology (ESCR) and the European Council of Nuclear Cardiology (ECNC). Eur J Nucl Med Mol Imaging 2011;38:201-12. [Crossref] [PubMed]
- Gaemperli O, Saraste A, Knuuti J. Cardiac hybrid imaging. Eur Heart J Cardiovasc Imaging 2012;13:51-60. [Crossref] [PubMed]
- Magalhães TA, Kishi S, George RT, et al. Combined coronary angiography and myocardial perfusion by computed tomography in the identification of flow-limiting stenosis - The CORE320 study: An integrated analysis of CT coronary angiography and myocardial perfusion. J Cardiovasc Comput Tomogr 2015;9:438-45. [Crossref] [PubMed]
- Koo BK, Erglis A, Doh JH, et al. Diagnosis of ischemia-causing coronary stenoses by noninvasive fractional flow reserve computed from coronary computed tomographic angiograms. Results from the prospective multicenter DISCOVER-FLOW (Diagnosis of Ischemia-Causing Stenoses Obtained Via Noninvasive Fractional Flow Reserve) study. J J Am Coll Cardiol 2011;58:1989-97. [Crossref] [PubMed]
- Min JK, Leipsic J, Pencina MJ, et al. Diagnostic accuracy of fractional flow reserve from anatomic CT angiography. JAMA 2012;308:1237-45. [Crossref] [PubMed]
- Nørgaard BL, Leipsic J, Gaur S, et al. Diagnostic performance of noninvasive fractional flow reserve derived from coronary computed tomography angiography in suspected coronary artery disease: the NXT trial (Analysis of Coronary Blood Flow Using CT Angiography: Next Steps). J Am Coll Cardiol 2014;63:1145-55. [Crossref] [PubMed]
- Douglas PS, De Bruyne B, Pontone G, et al. 1-Year Outcomes of FFRCT-Guided Care in Patients With Suspected Coronary Disease: The PLATFORM Study. J Am Coll Cardiol 2016;68:435-45. [Crossref] [PubMed]
- Patel MR, Dai D, Hernandez AF, et al. Prevalence and predictors of nonobstructive coronary artery disease identified with coronary angiography in contemporary clinical practice. Am Heart J 2014;167:846-52. e2.
- Patel MR, Peterson ED, Dai D, et al. Low diagnostic yield of elective coronary angiography. N Engl J Med 2010;362:886-95. [Crossref] [PubMed]
- Thom H, West NE, Hughes V, et al. Cost-effectiveness of initial stress cardiovascular MR, stress SPECT or stress echocardiography as a gate-keeper test, compared with upfront invasive coronary angiography in the investigation and management of patients with stable chest pain: mid-term outcomes from the CECaT randomised controlled trial. BMJ Open 2014;4:e003419. [Crossref] [PubMed]
- Dharampal AS, Papadopoulou SL, Rossi A, et al. Diagnostic performance of computed tomography coronary angiography to detect and exclude left main and/or three-vessel coronary artery disease. Eur Radiol 2013;23:2934-43. [Crossref] [PubMed]
- Berman DS, Kang X, Slomka PJ, et al. Underestimation of extent of ischemia by gated SPECT myocardial perfusion imaging in patients with left main coronary artery disease. J Nucl Cardiol 2007;14:521-8. [Crossref] [PubMed]
- Karlsberg RP, Budoff MJ, Thomson LE, et al. Reduction in downstream test utilization following introduction of coronary computed tomography in a cardiology practice. Int J Cardiovasc Imaging 2010;26:359-66. [Crossref] [PubMed]


